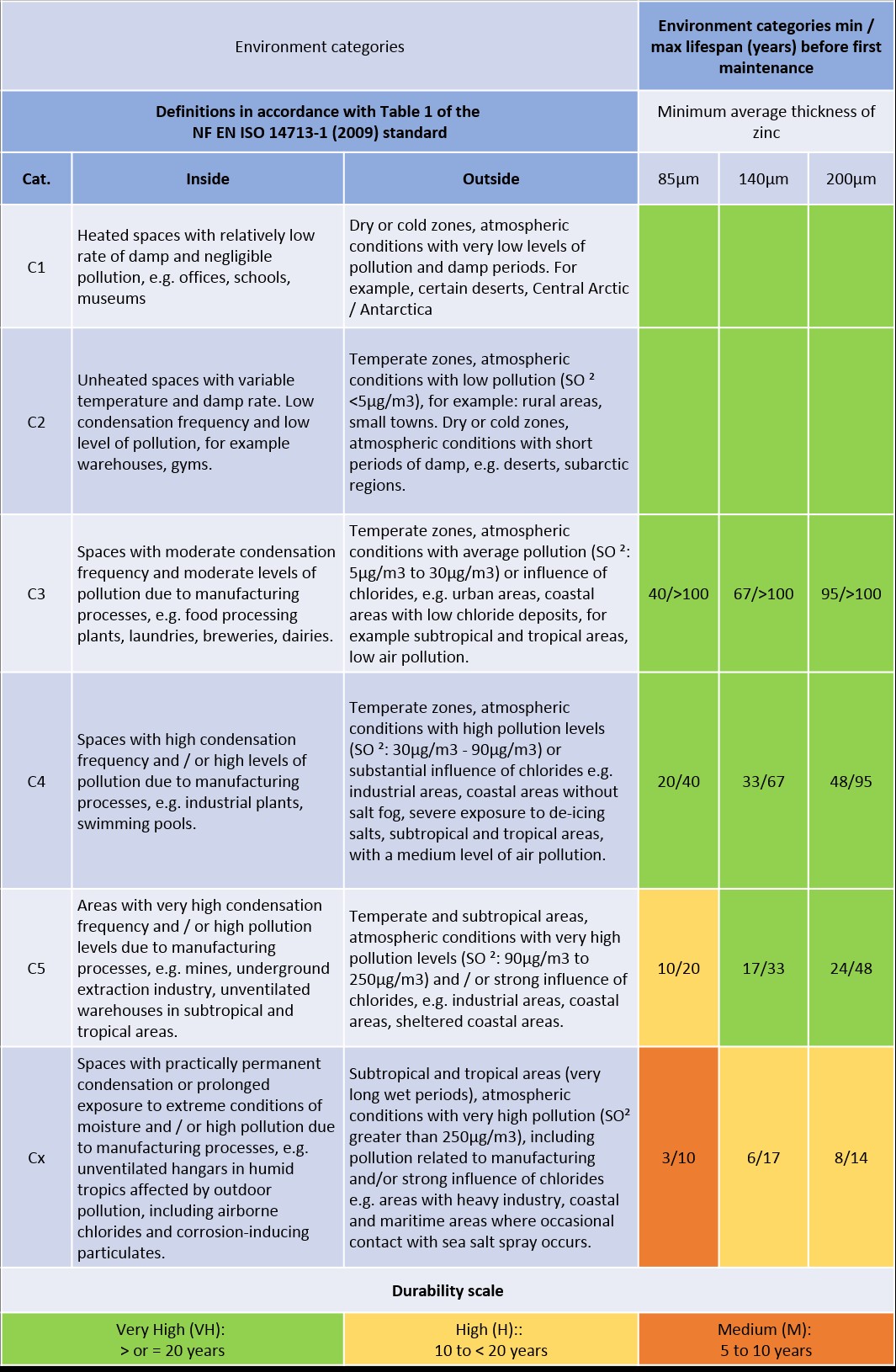
Galvanization lifespan
The galvanization lifespan depends on the thickness of the coating and the aggressiveness of the environment to which the galvanised parts are exposed.
Steels galvanized in image


CHOOSE A PLANT
The galvanization lifespan depends on the thickness of the coating and the aggressiveness of the environment to which the galvanised parts are exposed.
Steels galvanized in image



CHOOSE A PLANT